GIAPREZA in VASOPLEGIA
RESTORE MAP WITH A
REDUCED VASOPRESSOR LOAD
Trial Design
ATHOS-3: A landmark trial in vasoplegia1-3
GIAPREZA was studied in ATHOS-3, an international, randomized, double-blind, placebo-controlled trial of 321 patients with distributive shock who remained hypotensive despite fluid and vasopressor therapy.4,5
Slide table to view more
Treatment arms4 GIAPREZA (n=163) plus SOC vasopressors*,† Placebo (n=158) plus SOC vasopressors*,† | Primary endpoint4 An increase in MAP at hour 3 of at least 10 mmHg, or a MAP response of ≥75 mmHg, without an increase in baseline vasopressor dosing. |
Secondary endpoints4 - Mean change in cardiovascular SOFA score at hour 48 - Mean change in total SOFA score at hour 48 | |
Select additional endpoint4 All-cause mortality through day 28. |
Slide table to view more
| Select baseline patient characteristics (N=321)4 | GIAPREZA + SOC (n=163) | Placebo + SOC (n=158) |
|---|---|---|
| Median MAP, mmHg (IQR) | 66.3 (63.7–69.0) | 66.3 (63.0-68.3) |
| Median APACHE II score (IQR) | 27 (22-33) | 29 (22-34) |
| Cause of shock, sepsis (%) | 127 (77.9%) | 132 (83.5%) |
| Median vasopressor dose (NED), mcg/kg/min (IQR) | 0.33 (0.23-0.56) | 0.34 (0.23-0.56) |
| Vasopressin use during 6 hours before randomization (%) | 113 (69.3%) | 111 (70.3%) |
*Standard-of-care vasopressors included norepinephrine, epinephrine, dopamine, phenylephrine, and vasopressin.5
†Doses of GIAPREZA or placebo were titrated to a target MAP of ≥75 mmHg during the first 3 hours of treatment while doses of other vasopressors were maintained. From hour 3 to hour 48, GIAPREZA or placebo was titrated to maintain a MAP between 65 and 70 mmHg while reducing doses of other vasopressors.5
MAP Stabilization
3x more patients achieved and sustained target MAP when GIAPREZA was used with SOC4,5
Slide table to view more
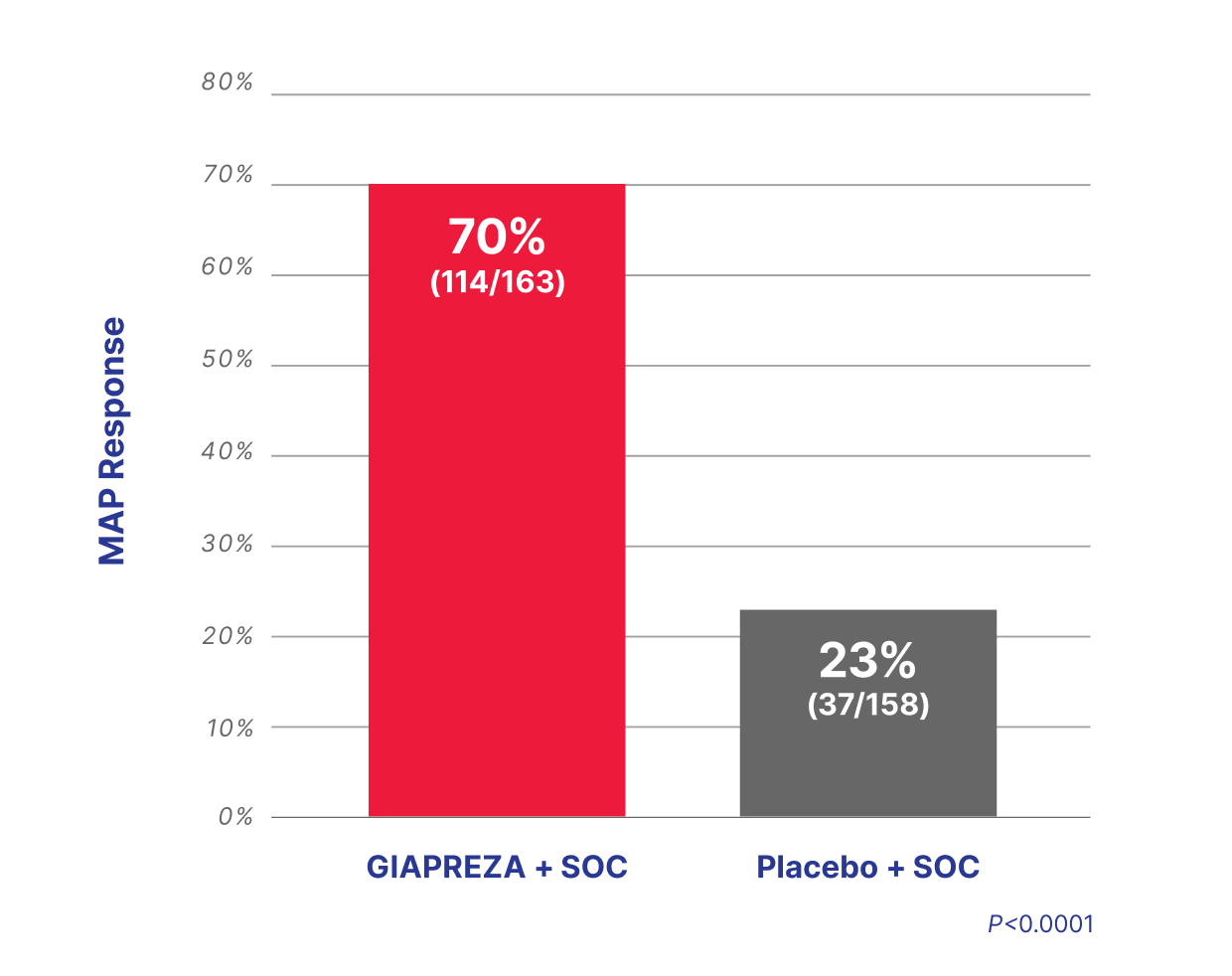
More vasoplegia patients achieved and
sustained target MAP with GIAPREZA plus SOC3
An ATHOS-3 post hoc subgroup analysis included 16 patients who were subsequently identified as having postoperative vasoplegia; 9 patients received ANG ll plus SOC and 7 patients received placebo plus SOC.3
Slide table to view more
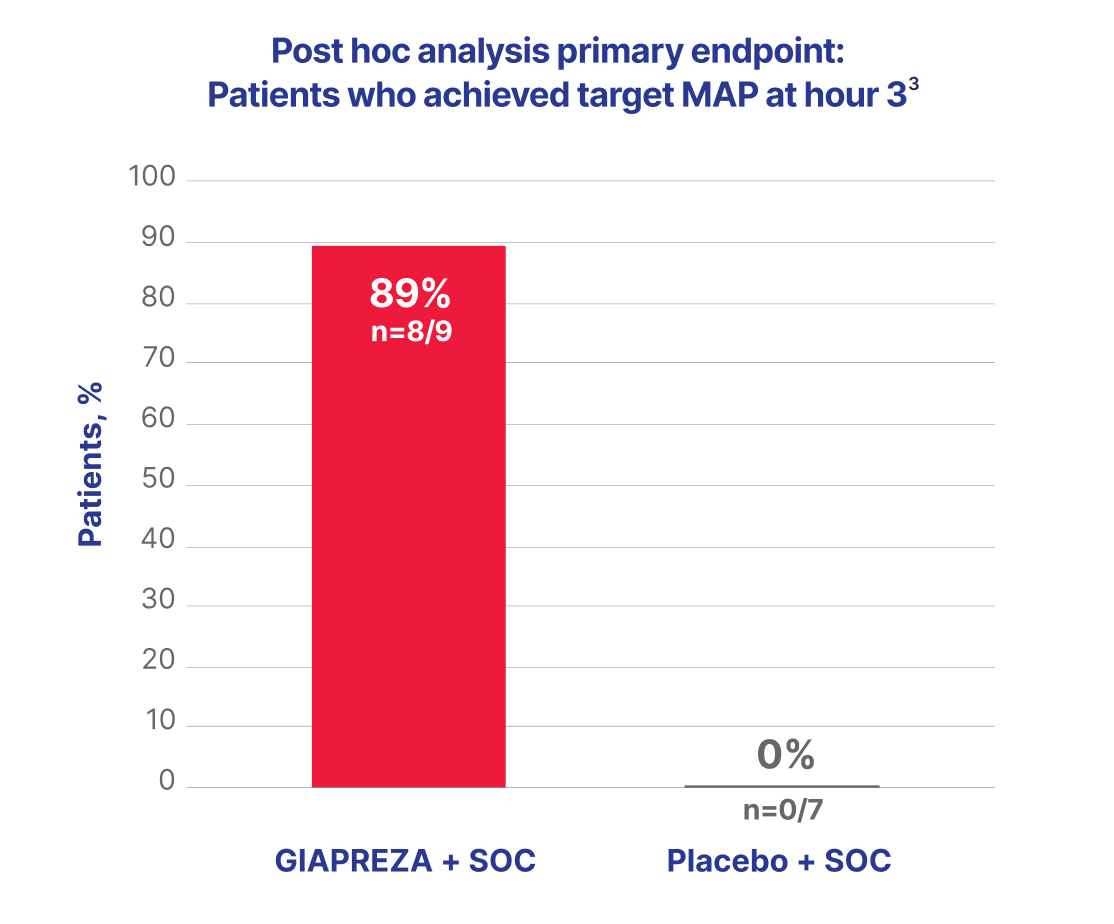
Responders in the ANG II group achieved target MAP within the first hour of treatment initiation, with a median MAP of 80.2 mmHg at hour 13


VASOPRESSOR LOAD
GIAPREZA demonstrated a vasopressor-sparing
effect in vasoplegia3
In an exploratory post hoc analysis of ATHOS-3, ANG II was associated with
a greater down-titration of SOC vasopressors vs placebo3
Slide table to view more
| Characteristic | GIAPREZA + SOC (n=9) | Placebo + SOC (n=7) | P Value |
|---|---|---|---|
| % change in NED at hour 12 Median (IQR) | -76.5 (-87.1 to -37.5) | 7.8 (-27.6 to 17.4) | 0.0013 |
In the GIAPREZA-treated group of the overall ATHOS-3 population, patients were not necessarily on maximum doses of other vasopressors at the time of randomization. The effect of GIAPREZA
when added to maximum doses of other vasopressors is unknown.5
Norepinephrine-equivalent dose (NED) is a standardized way to express the total vasopressor burden a patient is receiving—by converting all vasopressors into an equivalent dose of norepinephrine for comparison.6
Clinical Markers
GIAPREZA reduced renin and ANG I levels7
Select clinical outcomes of the subset of study patients in the
exploratory post hoc analysis of ATHOS-37
Slide table to view more
| GIAPREZA reduced renin and ANG I levels and was associated with better outcomes7 | GIAPREZA + SOC | Placebo + SOC |
|---|---|---|
| Median reduction in serum renin levels, % | 54.3 | 14.1 |
| Median reduction in serum ANG I levels, % | 39.7 | 7.0 |
| Outcomes: Patients above the study population median renin (median [IQR])7 | GIAPREZA + SOC | Placebo + SOC |
| 28-day mortality, % [median (IQR)]* | 51 (39–65) | 70 (59–80) |
| Ventilator liberation by day 7 (alive and ventilator-free), % (median [IQR])* | 28 (18–43) | 14 (8–25) |
| RRT liberation by day 7 (alive and RRT free), % (median [IQR])* | 43 (25–66) | 12 (5–27) |
| ICU discharge by day 28, % (median [IQR])* | 44 (32–58) | 22 (14–33) |
*Values represent outcomes in patients who had renin concentrations above the study population median.7
SAFETY
Safety and tolerability vs placebo and SOC in ATHOS-34
Slide table to view more
| GIAPREZA + SOC (n=163) | Placebo + SOC (n=158) | |
|---|---|---|
| Adverse event of any grade, % (n) | 87.1% (142) | 91.8% (145) |
| Adverse event leading to discontinuation, % (n) | 14.1% (23) | 21.5% (34) |
| Any serious adverse event with frequency ≥1% in either study group, % (n) | 60.7% (99) | 67.1% (106) |
Adverse reactions occurring in ≥4% of patients treated with GIAPREZA and ≥1.5% more often than in placebo‑treated patients included thromboembolic events, including arterial and venous thrombosis events (12.9% vs 5.1%), deep vein thrombosis (4.3% vs 0.0%), thrombocytopenia (9.8% vs 7.0%), tachycardia (8.6% vs 5.7%), fungal infection (6.1% vs 1.3%), delirium (5.5% vs 0.6%), acidosis (5.5% vs 0.6%), hyperglycemia (4.3% vs 2.5%), and peripheral ischemia (4.3% vs 2.5%).5
Thromboembolic events
- In ATHOS-3, serious adverse events (SAEs) were reported. The Prescribing Information grouped SAEs and adverse events, including thromboembolic events, as presented under a single category4,5
- The incidence of patients with a grade 3 or 4 (severe or life-threatening) thromboembolic event was 9 (5.5%) events in the GIAPREZA group vs 5 (3.2%) in the placebo group8
- In the GIAPREZA group, 12/25 (48%) thromboembolic events occurred within 5 days of randomization in comparison to 7/8 (87.5%) events in the placebo arm8
Use concurrent venous thromboembolism (VTE) prophylaxis.5
Fewer GIAPREZA-treated patients discontinued treatment vs placebo due to adverse events4
Fewer GIAPREZA-treated patients experienced adverse events and serious adverse events vs placebo4
Post hoc analysis of safety in patients with post-CPB vasoplegic shock in ATHOS-33
Occurrence of TEAEs was comparable between the ANG II and placebo groups.
There were no instances of venous or arterial thromboembolic events reported in patients with vasoplegia treated with ANG II. TEAEs from the ATHOS-3 post hoc analysis are summarized below.3
Slide table to view more
| n (%) | GIAPREZA + SOC (n=9) | Placebo + SOC (n=7) |
|---|---|---|
| All TEAE | 5 (55.6) | 7 (100.0) |
| Grade 3/4 TEAE | 3 (33.3) | 5 (71.4) |
| TEAE | 3 (33.3) | 5 (71.4) |
| TEAE resulting in discontinuation of study drug | 1 (11.1) | 1 (14.3) |
| Fatal outcome TEAE | 1 (11.1) | 1 (14.3) |
All patients in the angiotensin II treatment group were on anticoagulation compared to 6 (85.7%) patients in the placebo group.
Slide table to view more
*Instrumental ADL refers to preparing meals, shopping for groceries or clothes, using the telephone, managing money, and other such activities.9
†Self-care ADL refers to bathing, dressing and undressing, feeding self, using the toilet, and taking medications without being bedridden.9
Connect with a GIAPREZA sales
representative for information and support.
ADL=activities of daily living; ANG=angiotensin; APACHE 2=Acute Physiology and Chronic Health Evaluation II; ATHOS-3=Angiotensin II for the Treatment of High-Output Shock; CPB=cardiopulmonary bypass; IQR=interquartile range; MAP=mean arterial pressure; NED=norepinephrine-equivalent dose; RRT=renal replacement therapy; SOC=standard of care; SOFA=Sequential Organ Failure Assessment; TEAE=treatment-emergent adverse event.
References:
- Andrews L, Benken J, Benedetti E, et al. Effects of angiotensin II in the management of perioperative hypotension in kidney transplant recipients. Clin Transplant. 2022;36(9):e14754. doi:10.1111/ctr.14754
- National Center for Biotechnology Information. PubChem Compound Summary for CID 172198, angiotensin II. Accessed January 14, 2025. https://pubchem.ncbi.nlm.nih.gov/compound/Angiotensin-II-human
- Klijian A, Khanna AK, Reddy VS, et al. Treatment with angiotensin II is associated with rapid blood pressure response and vasopressor sparing in patients with vasoplegia after cardiac surgery: a post-hoc analysis of Angiotensin II for the Treatment of High-Output Shock (ATHOS-3) study. J Cardiothorac Vasc Anesth. 2021;35(1):51-58. doi:10.1053/j.jvca.2020.08.001
- Khanna A, English SW, Wang XS, et al. Angiotensin II for the treatment of vasodilatory shock. N Engl J Med. 2017;377(5):419-430. doi:10.1056/NEJMoa1704154
- GIAPREZA. Prescribing Information. La Jolla Pharmaceutical Company; 2021.
- Kotani Y, Di Gioia A, Landoni G, et al. An updated “norepinephrine equivalent” score in intensive care as a marker of shock severity. Crit Care. 2023;27(1):29. doi:10.1186/s13054-023-04322-y
- Bellomo R, Forni LG, Busse LW, et al. Renin and survival in patients given angiotensin II for catecholamine-resistant vasodilatory shock. A clinical trial. Am J Respir Crit Care Med. 2020;202(9):1253-1261. doi:10.1164/rccm.201911-2172OC
- Data on File. Innoviva Specialty Therapeutics; 2023.
- Common Terminology Criteria for Adverse Events (CTCAE) Version 4.0. US Department of Health and Human Services, National Institutes of Health, National Cancer Institute. Published May 28, 2009. Updated June 14, 2010. Accessed December 12, 2025. https://evs.nci.nih.gov/ftp1/CTCAE/CTCAE_4.03/CTCAE_4.03_2010-06-14_QuickReference_8.5x11.pdf
IMPORTANT SAFETY INFORMATION
Indication
GIAPREZA® (angiotensin II) increases blood pressure in adults with septic or other distributive shock.
Contraindications
None.
Warnings and Precautions
The safety of GIAPREZA was evaluated in 321 adults with septic or other distributive shock in a randomized, double-blind, placebo-controlled study, ATHOS-3. There was a higher incidence of arterial and venous thrombotic and thromboembolic events in patients who received GIAPREZA compared to placebo-treated patients in the ATHOS-3 study (13% vs. 5%). The major imbalance was in deep venous thromboses. Use concurrent venous thromboembolism (VTE) prophylaxis.
Adverse Reactions
The most common adverse reactions reported in greater than 10% of GIAPREZA-treated patients were thromboembolic events. Adverse reactions occurring in ≥4% of patients treated with GIAPREZA and ≥1.5% more often than placebo-treated patients in the ATHOS-3 study were thromboembolic events (including deep vein thrombosis), thrombocytopenia, tachycardia, fungal infection, delirium, acidosis, hyperglycemia, and peripheral ischemia.
Drug Interactions
Angiotensin converting enzyme (ACE) inhibitors may increase response to GIAPREZA. Angiotensin II receptor blockers (ARBs) may reduce response to GIAPREZA.
You are encouraged to report negative side effects of prescription drugs to the FDA.
To report SUSPECTED ADVERSE REACTIONS, please contact:
Before administering, please see the Full Prescribing Information for GIAPREZA.