Using Giapreza
Dosing and administration
DOSING
Flexible and easily titratable for rapid MAP control
1
INITIATE
- The recommended starting dose of GIAPREZA is 20 ng/kg/min (0.02 mcg/kg/min)
2
access
- The median time to reach target MAP was 5 minutes
- The median dose of GIAPREZA was 10 ng/kg/min at 30 minutes
3
optimize
- Titrate GIAPREZA as frequently as every 5 minutes by increments of up to 15 ng/kg/min as needed to achieve or maintain target blood pressure
- The half-life of GIAPREZA is less than 1 minute
- Once the underlying shock has sufficiently improved, down‑titrate every 5 to 15 minutes in increments of up to 15 ng/kg/min, based on blood pressure
Doses as low as 1.25 ng/kg/min
may be used
During the first 3 hours, the maximum dose should not exceed 80 ng/kg/min
Maintenance dose should not exceed 40 ng/kg/min
To learn more about dosing, download a Dosing and Administration Guide
PREPARATION
Available in 0.5 mg/ml and 2.5 mg/ml vial options
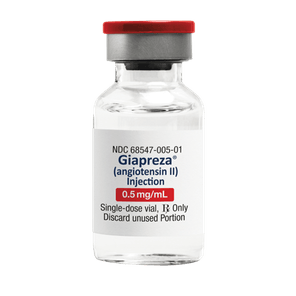
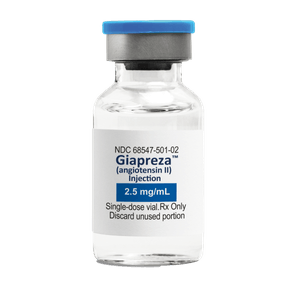
GIAPREZA injection is a clear, aqueous solution supplied in a carton with a 1 mL single-dose vial containing 0.5 or 2.5 mg angiotensin II as a sterile liquid*
- Inspect for particulate matter and discoloration prior to administration
- Dilute the appropriate amount of GIAPREZA in a normal saline (0.9% sodium chloride) infusion bag to achieve the desired final concentration of 5,000 ng/mL or 10,000 ng/mL
*Remember that GIAPREZA dosage is measured in nanograms (ng), not micrograms (mcg)
ADMINISTRATION & STORAGE
How to administer and store
Administration
Administer GIAPREZA
via continuous intravenous infusion
Administration through a central venous line is recommended
Monitor blood pressure closely after GIAPREZA initiation
Storage
- GIAPREZA vials should be stored in the refrigerator between 2°C to 8°C (36°F to 46°F)
- Diluted solution may be stored at room temperature or under refrigeration for up to 24 hours, after which it should be discarded
- Discard vial and any unused portion of the drug product after use
WASTAGE PRODUCT REPLACEMENT PROGRAM
Innoviva Specialty Therapeutics will replace eligible vials of GIAPREZA free of charge in the event that GIAPREZA vials are spoiled, or are otherwise unable to be administered.
For eligibility criteria and instructions on initiating replacement requests, please fill out this form and refer to the Terms and Conditions below.
If you have any questions, please contact fieldops@ljpc.com.
LIMITED WARRANTY PROGRAM
Innoviva Specialty Therapeutics will replace eligible vials of GIAPREZA free of charge in the event that a patient receiving GIAPREZA does not reach an increase in mean arterial pressure (MAP) of 10 mmHg, or reach a MAP of 65 mmHg, within the first three hours of initiation.
For eligibility criteria and instructions on initiating warranty claims, please fill out this form and refer to the Terms and Conditions below.
If you have any questions, please contact warranty@ljpc.com.
Connect with a GIAPREZA sales
representative for information and support.
Important Safety Information
Indication
GIAPREZA® (angiotensin II) increases blood pressure in adults with septic or other distributive shock.
Contraindications
None.
Warnings and Precautions
The safety of GIAPREZA was evaluated in 321 adults with septic or other distributive shock in a randomized, double-blind, placebo-controlled study, ATHOS-3. There was a higher incidence of arterial and venous thrombotic and thromboembolic events in patients who received GIAPREZA compared to placebo-treated patients in the ATHOS-3 study (13% vs. 5%). The major imbalance was in deep venous thromboses. Use concurrent venous thromboembolism (VTE) prophylaxis.
Adverse Reactions
The most common adverse reactions reported in greater than 10% of GIAPREZA-treated patients were thromboembolic events. Adverse reactions occurring in ≥4% of patients treated with GIAPREZA and ��≥1.5% more often than placebo‑treated patients in the ATHOS-3 study were thromboembolic events (including deep vein thrombosis), thrombocytopenia, tachycardia, fungal infection, delirium, acidosis, hyperglycemia, and peripheral ischemia.
Drug Interactions
Angiotensin converting enzyme (ACE) inhibitors may increase response to GIAPREZA. Angiotensin II receptor blockers (ARBs) may reduce response to GIAPREZA.
You are encouraged to report negative side effects of prescription drugs to the FDA.
To report SUSPECTED ADVERSE REACTIONS, please contact:
Before administering, please see the Full Prescribing Information for GIAPREZA.